This may have been responsible for the resulting angioedema contralateral to the inciting stroke. In our patient, the right M2 segment of the MCA was affected which may have resulted in autonomic dysfunction of the insular cortex. Given that angioedema was noted contralateral to the location of our patients stroke we postulate that this is likely due to ischemic changes. In this same study by Hill et al., 7 out of the 9 patients were found to have angioedema on the contralateral side of the ischemic stroke as was seen in our patient. In this study, 5.1% were found to have angioedema secondary to IV alteplase administration. described a 5-year study involving 176 patients treated with IV alteplase for acute stroke. The documented incidence of angioedema secondary to tPA administration increases with the use of ACE inhibitor. Angioedema secondary to tPA administration for acute stroke is estimated at 0.2–5.1%. Several studies have shown that the incidence of oral lingual angioedema with tPA administration for stroke may be higher than previously understood. Angioedema secondary to tPA administration is a rare side effect with an estimated incidence of 0.02% in patients being treated with alteplase for acute MI. Mild cases may respond to antihistamines, while more severe cases may require corticosteroids, epinephrine, and even bradykinin antagonists like Icatibant. Treatment of angioedema is driven by the inciting cause. DiscussionĪngioedema secondary to ACE inhibitor treatment is well documented with an incidence of 0.1%–2%. Patient was stabilized and admitted to neurology intensive care unit for close observation after tPA administration and for initiation of stroke risk factor reducing therapies. Patient was found to have patent airway throughout the episode, so decision was made to hold treatment and monitor closely. Given lack of concerns for airway obstruction, patient was closely observed in the emergency room setting. A diagnosis of oral lingual angioedema secondary to tPA administration was made. Patient denied SOB or difficulty breathing and had SaO2 92%. Inspection of the oral cavity revealed no trauma to the tongue or hematoma. Reassessment revealed acute onset of swelling to the patient’s left side of her tongue contralateral to the location of the stroke (see Figure 1). Within 20 minutes of onset of tPA administration, patient started to complain of facial swelling. Patient was given IV tPA calculated from weight with a bolus dose of 7 mg and an infusion dose of 62.85 mg. CT head showed a tapered occlusion of the superior division of the right M2 segment, approximately 1.4 cm distal to the origin with resultant ischemic stroke of right M2 segment of the MCA. NIHSS assessment was performed and found to be 6: 2 points for partial facial paralysis, 1 point for left arm drift, 2 points for sensory loss in left face, arm, and leg, and 1 point for mild slurred speech. On physical exam, patient had left sided facial droop with resulting slurred speech. Review of patients prior to admission medication showed that patient was taking Lisinopril 40 mg QD. On presentation, patient’s glucose was 123. Upon arrival by EMS, the patient was brought directly to CT for imaging where the neurology stroke team was awaiting to assess the patient. Patient is a 62-year-old female who presented to the emergency department as a stroke call due to symptoms of left sided weakness, facial droop, and slurred speech. We present the case of a 62-year-old female who presented with signs and symptoms of acute stroke and was treated with IV tPA that resulted in self-limiting angioedema.

The incidence of angioedema increases in patients who also take an ACE inhibitor.
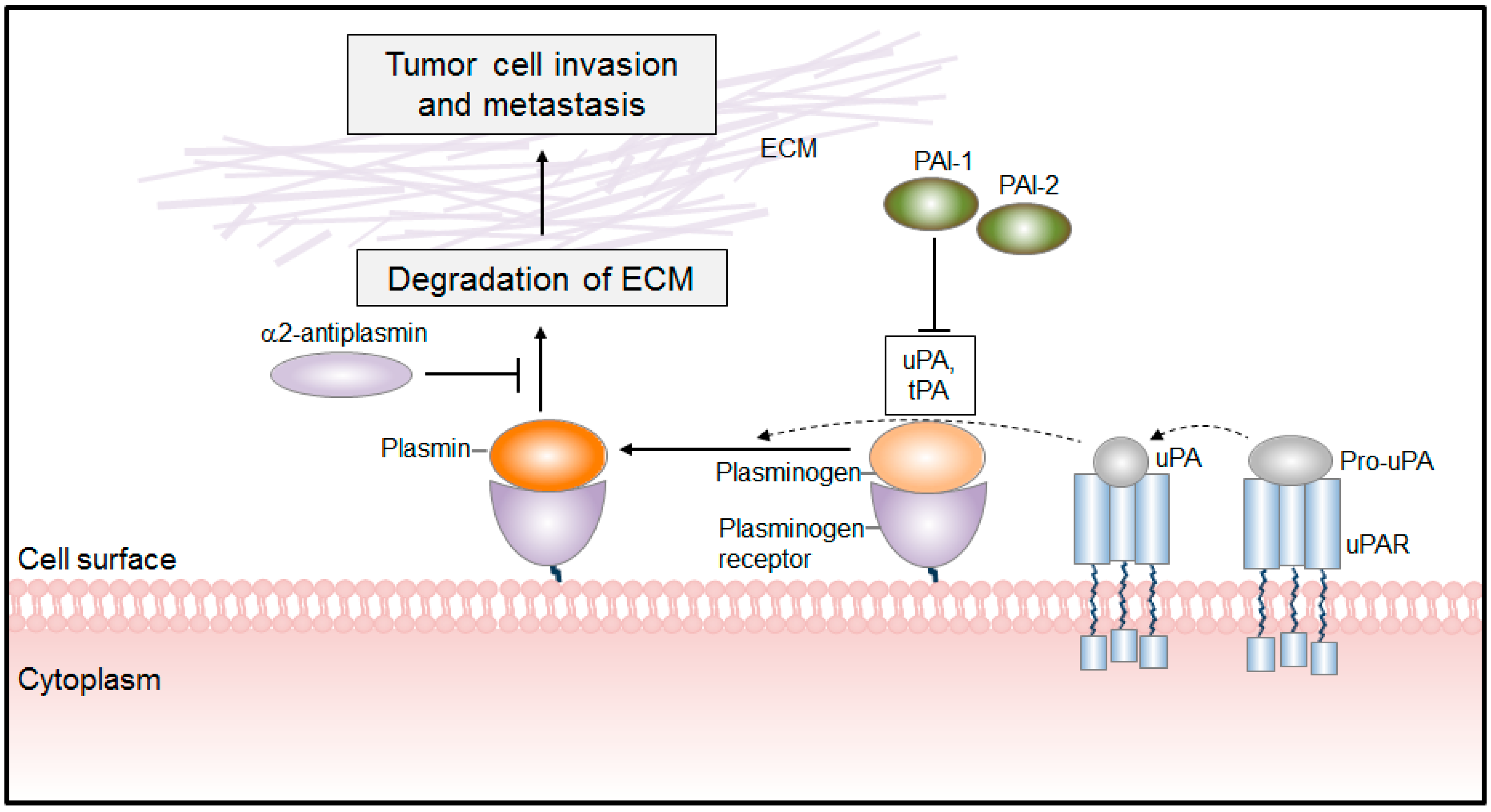
Common side effects of IV tPA include intracerebral hemorrhage, systemic hemorrhage, and rarely angioedema.

In 1996, IV tPA was approved for treatment of acute ischemic stroke and since then has been the standard of treatment for patients experiencing ischemic stroke.
IntroductionĪpproximately 795,000 Americans experience a stroke each year with 87% of these being ischemic. This is of particular importance for practitioners in the acute care setting. Given the frequent and widespread use of tPA, awareness of the rare life-threatening side effects is paramount. We present the case of a patient treated for ischemic stroke who developed angioedema and discuss the etiology and risk factors for this rare, but dangerous side effect. One rare and potentially dangerous side effect is angioedema. In a rare set of patients treated with IV tPA, side effects can be seen. In the majority of patients, this offers an effective and often life-prolonging treatment in the acute setting.
#TISSUE PLASMINOGEN ACTIVATOR ACTIVATOR#
IV tissue plasminogen activator (tPA) is the treatment of choice for ischemic strokes that present within the treatment window.
